- A substance is a form of particles that can be group into more specific classes.
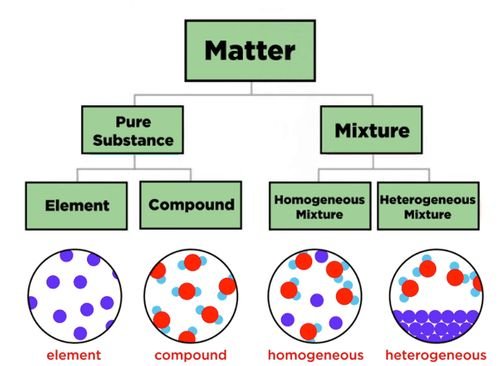
- There are two main forms of substances, pure substances and mixtures.
- A chart showing the different groupings of substances.
Pure Substances
- Pure substances are either raw elements or compounds of elements, such as carbon dioxide or water.
- Compounds are made of two or more elements, bonded together.
- Compounds can be ionic, such as NaCl, or covalent, like H₂0 for example.
- Covalent compounds are always formed from non-metals, with a few exceptions such as silicon.
- Ionic compounds are formed between metals and nonmetals.
- Compounds can only be separated through chemical methods (chemical reactions).
Mixtures
- Mixtures consist of two or more pure substances.
- The ingredients of the mixture can be separated from each other by physical methods such as evaporation, filtration, or distillation.
- Mixtures can be homogenous, meaning that it is full mixed and the different substances can't be differentiated.
- The different substances in homogenous mixtures can't even be seen under microscopes, as you'd need to see the individual atoms to tell the difference apart.
- For example salt dissolved in water.
- Otherwise mixtures can be heterogenous, meaning that the substances can easily be differentiated.
- For example sand in water.
- Heterogenous mixtures can be often be separated with filters, whereas homogenous mixtures often require more elaborate methods to separate.
- Miscibility is how easily a substance forms mixtures. A miscible substance can mix easily, whereas an immiscible substance usually won't form mixtures.
- Although the phrase is commonly used for liquids, it can also be applicable to gases and solids.
Phases
- A phase is a region of state in a mixture with precise boundaries.
- The term phases can usually be used interchangeably with states of matter, however not that in some cases the same phases might be immiscible.
- For example, how a solid and a liquid won't mix; it means they are in different phases (a liquid phase and a solid phase).
- The same can apply for the same states of matter, such as the boundary between cooking oil and water.
- When a substance is in one phase(meaning the mixture has fully mixed), it is homogenous.
- For instance, milk is an example of a homogenous mixture, as you can't tell apart the different substances in a glass of milk.
- In a homogenous liquid solution, the solute and solvent mix perfectly.
- In a saturated solution, more solute cannot be dissolved by the solvent at the current temperature.
Suspensions, Emulsions and Colloids
Suspensions
- A suspension is a fluid which contains solid particles with a size of over 1 micrometer (or 1000 nanometers).
- This means the solid particles are too large to float and eventually fall to the bottom of the container.
- Thus suspensions aren't solutions, as both substances have distinct and easily noticeable phases.
Colloids
- The opposite of a suspension is a colloid.
- In a colloid particles are smaller than 1 micrometer, and thus float in the liquid.
- Although colloids may appear like a true solution, they are still heterogenous and thus not a true solution.
Emulsions
- An emulsion is similar to a colloid, however the mixture only contains liquids.
- It is when two liquids that normally don't mix are in a mixture (similar to the solid particles floating around in a colloid, but liquid).
- Although emulsions, like colloids, might look homogenous on a macroscopic level, they can be differentiated on a microscopic level and thus not true solutions.
