- Water can exist on Earth simultaneously in all three states of matter.
- It exists as liquid water, water vapor and ice.
- Ice floats on water due to its lower density.
- Water does not dissolve cooking oil.
- This is because water is denser than nonpolar cooking oil, which is why the oil remains in its own layer on the top of the water.
Water as a Solvent
- A water soluble dye dissolves in water.
- Due to thermal movement it mixes evenly with the water.
- Once the dye and water are mixed, they can't be easily separated.
- This movement is called diffusion.
- Diffusion is the movement of a substance from a place of higher concentration, to a place of lower concentration.
- For example if you spray perfume then the perfume diffuses into the room as it moves from its concentrated form in the bottle to encapsulate the entire room.
- Water dissolves polar molecules because hydrogen bonds are formed between polar molecules and water molecules, causing the substance to dissolve into the water.
- Many ionic compounds are easily soluble in water.
- Nonpolar substances don't dissolve in water, but instead can be dissolved in nonpolar solvents such as gasoline.
- Since the molecules of substances in solutions are externally uncharged, they do not conduct electricity.
- Acids are usually covalent compounds, and thus when they dissolve in water, the reaction results into an aqueous solution with positive hydrogen ions (H+) which conduct electricity.
- The ions conduct electricity, and therefore so do acids in aqueous solutions.
- Bases also dissolve in water as they release negatively charged hydroxide ions (OH-).
- Both the H+ and OH- ions act as charge carriers and thus conduct electricity.
- The dissolution of ionic compounds in water is called hydration.
- During hydration, aqua ions are formed, which then bond to water molecules due to dipole-dipole forces.
- An ion-dipole force is a intermolecular force (which are weak bonds) but it is stronger than intermolecular forces between molecules.
Osmosis
- Osmosis is the movement of water across a semi-permeable membrane from an area of high water concentration to an area of lower water concentration.
- It is an important process in biology.
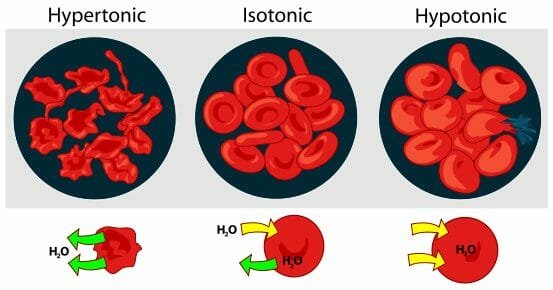
- A diagram showing the effect of osmosis on red blood cells.
Surface Tension
- All liquids have some level of surface tension, although water has the highest due to its ability to form hydrogen bonds.
- Water molecules form a sort of layer on the surface of water.
- This is called surface tension, and it acts as a weak barrier.
- It is too weak for us to considerably notice it.
- It is why ants can drown in water droplets, as they can get stuck from the surface tension.
- Its also why water striders can skim across water without sinking.
- Surface tension is also the cause for the phenomenon of capillary action.
Image Sources
https://biologydictionary.net/osmosis/