Properties of Metals
- Metals have lose, free moving valence electrons.
- They are called delocalized electrons.
- Metals can thus form metal lattices.
- They are compromised of one or more types of metal.
- Alloys such as bronze, steel and brass are formed from several types of metal, and the different sizes of atoms can help make a stronger lattice.
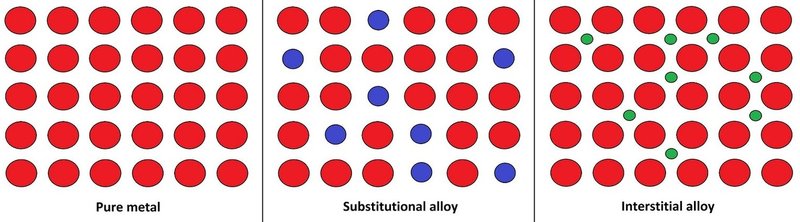
- An illustration of metal lattices, of both normal metals and alloys.
- Most of the elements in the periodic table are metals.
- Metals are great conductors of heat and electricity.
- Metals can be shaped without breaking.
- The cation layers simply slide past each other without separation.
- Metals are naturally sonorous.
- Metals are generally durable and strong.
- Metals are also shiny and opaque.
Ways of Differentiating Metals
- Form in room temperature: Mercury is a liquid, other metals are solids.
- Radioactivity: some metals are radioactive (although there aren't many in nature as most radioactive metals have decayed).
- Alkali: Some metals are highly reactive and alkaline, whereas others, such as noble metals (gold, silver and copper for example) are barely reactive.
Properties of the Metal Lattice
- The metal lattice is held together by a metal bond.
- The metal lattice is a strong bond, which is what gives metals their structural durability.
- In the solid state, metals form lattice: a regular repeating structure which is held together by a metal bond.
- This lattice causes the melting temperatures of metals to be rather high (other than mercury).
- The lattice is held together with strong bonds which are hard to break.
- Metals are good conductors of heat and electricity because of their lattice.
- Heat causes thermal vibration in the tightly-knit lattice structure, which flows along the metal. This causes effective conduction.
- Due to their lattice, which can shift without breaking, metals are ductile and malleable. This means that they can be shaped and bent without breaking.
- Because of the delocalized electrons, the repulsive nature of the cations does not cause the structure of the metal lattice to break when shifted.
- Light is reflected from a smooth metal surface and the metal appears shiny.
- When visible light hits the surface of a metal, it cause the delocalized electrons to excite.
- When the excited state is released, the energy is released as visible light, which we see as reflections on the surface of the metal.
- Part of the light is observed in the dense metal lattice, this causes the opacity of metals, as wavelengths of visible light do not pass through this dense lattice.
