Ionic Bonding
- The structural parts of substances are connected to each other by various bonds.
- Examples of strong bonds are: Ionic bonds, metallic bonds and covalent bonds.
- Ionic bonds are the strongest type of atomic bond.
- Ionic bonds form between metals and nonmetals.
- An ionic bond is an attraction between ions of different signs.
- This is because negative and positive charges attract each other, causing the ions to form bonds.
- Ionic bonds are held together by their strong opposite electrical charges and they the difference of electronegativity between atoms in the compound is greater than 1.7.
- Keep in mind that ionic compounds have no partial charges.
Formation of Ions
- Atoms always want to have full shields of electrons, and often give up or take electrons to make a full shield.
- When a metal ion gives up an electron or electrons, it forms a positive ion.
- Alkali metals and alkali earth metals are very prone to giving up electrons as they only have 1 or 2 valence electrons.
- Some other metals like copper, gold and silver are very unreactive.
- Normally the charge is balanced, however when giving up electrons it becomes positively charged.
- Thus they become positive ions, which are called cations.
- When a nonmetal receives an electron or electrons, it forms a negative ion, called an anion.
- By taking negative electrons, the charge of an atom becomes negative, and thus unbalanced.
- For example oxygen has 8 electrons, meaning only 1 of its shields are complete, whereas its second shield still needs 2 electrons to be a full octet.
- This is why oxygen forms negative ions.
- Halogens and chalcogens are the most prone to reacting.
- Noble gases however practically never react as they have full valence shields.
Ionic Charges
- Ionic charge can be inferred from the difference between protons and electrons.
- For example, oxygen has 8 electrons and 8 protons. Thus when oxygen receives an additional 2 electrons, it has 10 electrons.
- By calculating we can infer that: 8 - 10 = -2
- This means that oxygen ions have a charge of negative 2.
Salts
- A salt is an ionic compound, or a salt, consists of ions.
- Not to be confused with NaCl, which is sea salt or table salt.
- NaCl is a salt, but there are many other salts than just NaCl.
- Salts are compounds formed by metals and nonmetals.
- Cations and anions are bind to each other with ionic bonds.
Formulating Ionic Compounds
- Complete ionic compounds should have a total charge of 0, this is because the cations and anions in the compound should cancel each other out.
- In the formula, cations are written first, and anions are written next.
- The same applies to the name.
- For example Na+ and Cl- form NaCl.
- The relative number of ions is indicated by subscripts.
- Names of metals in ionic compounds remain the same, (Magnesium, Sodium, Lithium, etc.) whereas nonmetals often have their names changed.
- Such as Halogens, whose names end in "-ide" when in an ionic compound.
- For example: Chlorine -> Chloride, Fluorine -> Fluoride, Sulphur -> Sulphide, Oxygen -> Oxide.
- After the metal you might see (I), (II), (III) and so forth, which indicates the positive charge of the metal.
Properties of Ionic Compounds
- Due to their polarities, ionic compounds form an ionic lattice while in a solid state.
- An ionic lattice is a three-dimensional, repeating structure that is formed by ionic bonding.
- Ionic lattices have an alternating pattern, as the opposite sides of an ionic compound attract one another.
- Ionic compounds have high melting points.
- Ionic compounds are hard but brittle. This is because they are in a lattice, however the lattice can split on impact. This is because the ions of the same charge repel each other when the lattice is shifted.

- An image showing the ionic lattice formed by sodium chloride, or NaCl.
- Ionic compounds conduct electricity when dissolved in an aqueous solution, but not in a solid state.
- As a solid, salts can't conduct electricity due to being tightly together and having a neutral charge. However in water they separate into their ions which are charged.
Crystallization of Ionic Compounds in Water
- When water evaporates from the salt solution, some of the water molecules may remain in the ion lattice.
- The water is called water of crystallization.
- The water of crystallization is water in an ion lattice.
- Crystal water is indicated in the formula of an ionic compound by a dot.
- For example: CuSO₄.5H₂O
- This would mean that the crystallized copper sulphate would have 5 water molecules per copper sulfate molecule. It is called copper sulphate pentahydrate, the prefix penta- meaning 5.
The Sizes of Ions
- Atoms sizes can vary depending on their type of ion.
- Cations are smaller than their non-ionic counterparts because they've given up an electron or multiple, reducing the size of their electron shields.
- This is the opposite of anions, which have larger sizes than their neutral counterparts because they have additional electrons, which increase their size.
- This effect scales, as the greater the positive charge of a cation, the smaller it's radius.
- The opposite applies to anions as more negatively charged anions are larger.
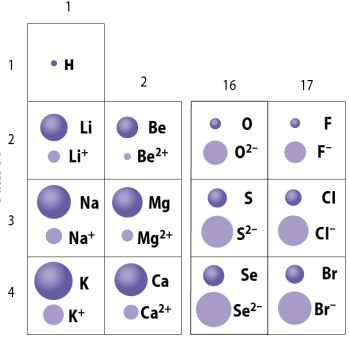
- A chart showing the various sizes of ions.
Image Sources
https://theory.labster.com/crystal-lattice/